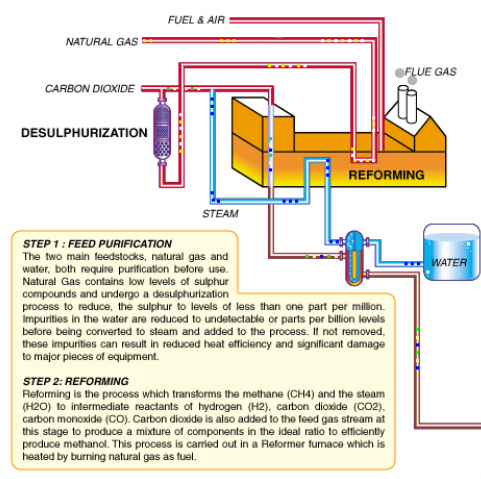
PRODUCTION OF METHANOL
Methanol is a very useful form of alcohol, commonly found in fuels, as well as formaldehyde. The synthesis of methanol is carried out by reacting hydrogen gas with carbon monoxide gas. The hydrogen and carbon monoxide gases are obtained through the oxidation of methane. In this reaction, methane is reacted with oxygen at a relatively low temperature(1 ATM), causing the reaction to favour this side with more moles, in this case the products. This reaction is effective for obtaining carbon monoxide and hydrogen gas because is delivers the 1:2 ration of carbon monoxide to hydrogen gas that is required for the synthesis of methanol. This reaction is represented by the equation below:
CH4(g) + 1/2O2(g) → CO(g) + 2H2(g)
Once the carbon monoxide and hydrogen gas are obtained from the oxidation of methane, they are reacted together to form methanol gas. The reaction is represented by the equation below:
CO(g) + 2H2(g) → CH3OH(g)
Methanol is second in the world in amount shipped and transported around the globe every year and approximately At room temperature, it is a polar liquid, and is used as an antifreeze, solvent, fuel, and as a denaturant for ethanol. It is also used for producing biodiesel via transesterification reaction.
CH4(g) + 1/2O2(g) → CO(g) + 2H2(g)
Once the carbon monoxide and hydrogen gas are obtained from the oxidation of methane, they are reacted together to form methanol gas. The reaction is represented by the equation below:
CO(g) + 2H2(g) → CH3OH(g)
Methanol is second in the world in amount shipped and transported around the globe every year and approximately At room temperature, it is a polar liquid, and is used as an antifreeze, solvent, fuel, and as a denaturant for ethanol. It is also used for producing biodiesel via transesterification reaction.